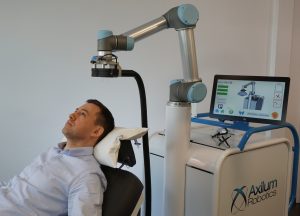
Strasbourg, February 21th 2019 – Axilum Robotics, specializing in the development of medical robots, announces that, 2 weeks after the CE mark, the Company has received 510(k) clearance from the U.S. Food and Drug Administration to market the TMS-Cobot TS MV, indicated for the spatial positioning and orientation of the treatment coil of the MagVenture TMS Therapy system.