TUS-Cobot is a robotic device designed for Transcranial Ultrasound.
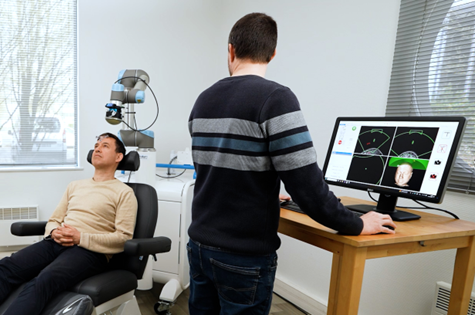
Overview of TUS-Cobot piloted by TUS-Navigator
TUS-Cobot is neither CE marked nor FDA cleared. It is a device for investigational use only.
Key features :
- Use Axilum Robotics Cobot platform that is CE marked and FDA cleared for Transcranial Magnetic Stimulation application.
- Collaborative robotic technology for safety.
- Compatible with several transducers via a specific adaptation kit. The adaptation kit comprises a 3D printed part into which the compatible TUS transducer, a water bladder coupling system with input and output and a third hose for water pressure measurement are inserted. The skin contact is made through a disposable flexible membrane which is user-replaceable.
- Pilotable by Axilum Robotics’ TUS-Navigator and compatible neuronavigation systems (Localite, Brainsight ongoing), with TUS enabled functionalities.
- Control of:
- Distance between the target and the head to maintain desired position,
- Relative contact pressure in the water bladder when applied on the head for additional safety.
- Appropriate coupling with coverage of all the surface of the transducer by filling the water bladder with more or less of degassed water depending on the desired thickness (relative pressure measurement).
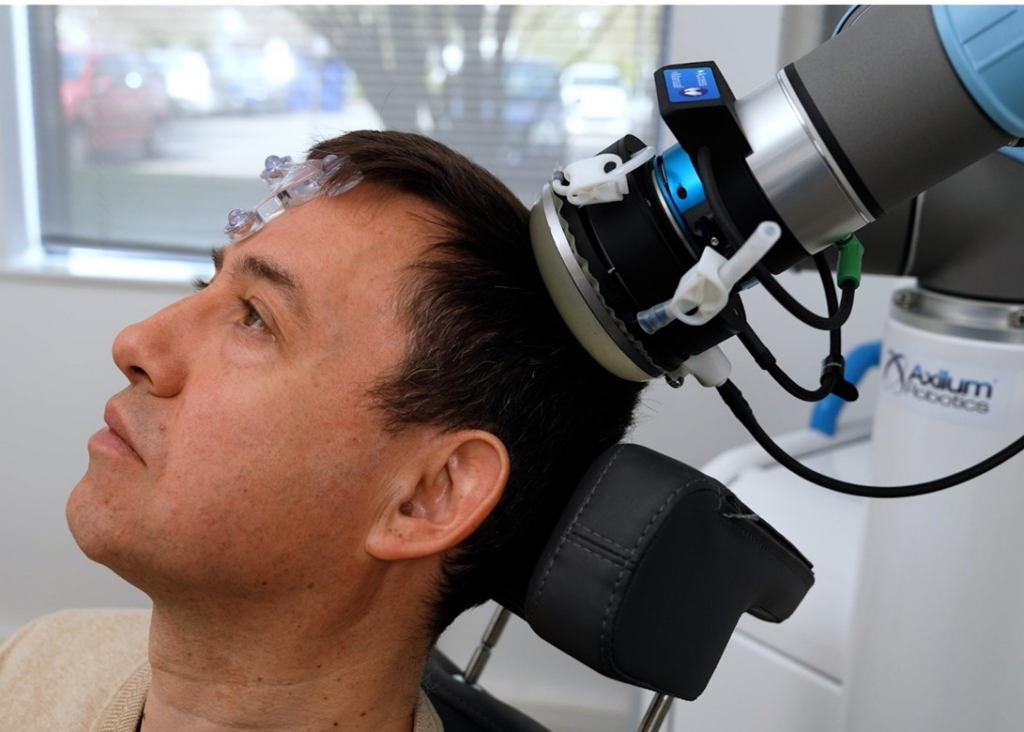
Close view on the transducer applied to a patient’s head
- Spherical trajectories to avoid head collision, and real-time contact management for safety.
- Head motion compensation.
- Suitable for multiple targets procedures.
- Double TUS and TMS compatibility: with a new tool holder and the QuickChanger device it is also now possible to easily and quicky mount either a compatible TMS coil or a compatible TUS transducer, both equipped with their adaptation kit. Click for a demo video.
